|
The oxygen, on the other hand, being more electronegative, likes to share electrons to create a more stable electron configuration. 
In every molecule of water (H 2O), the hydrogen is less inclined to keep its electron tightly bound. Electronegativity also is the source for the occurrence of electrical polarity in substances. 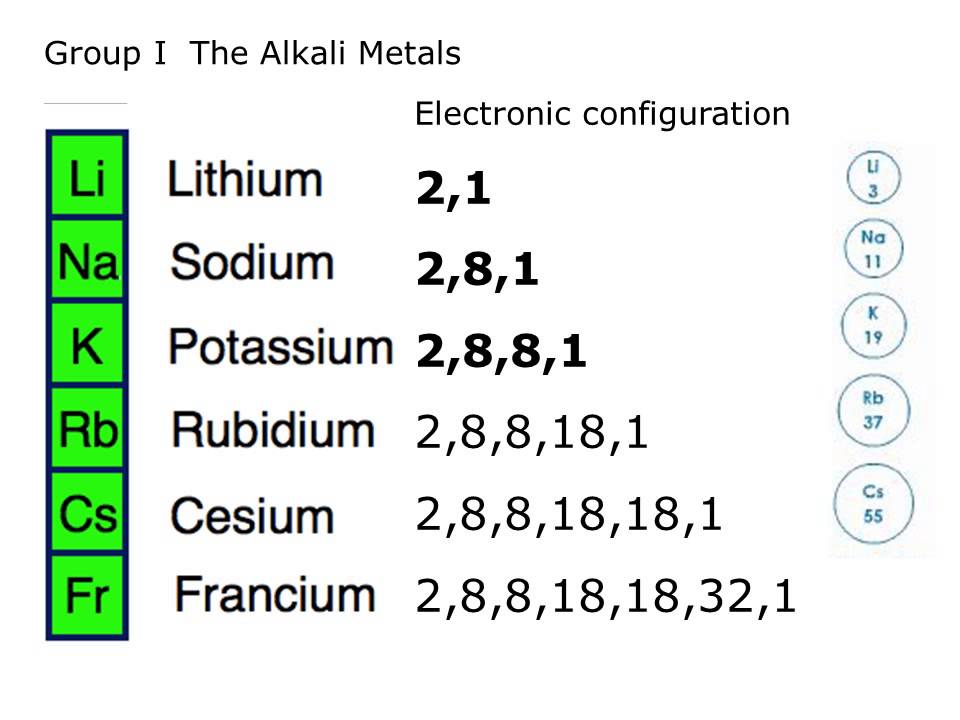
hydrofluoric acid is among the strongest acids known to mankind, where sodium hydroxide is one of the strongest bases - as an example. The halogens are reactive for the opposite reason – they need to attract that electron to obtain a more stable electron-configuration. The alkali metals are the most eager to lose an electron (they have the lowest electronegativity in the row) making them very reactive with water. For the latter reason, the tendency to lose an electron to form a covalent bond decreases and turns into the tendency of needing one or more electrons to form a stable covalent bond, producing a more stable chemical substance or molecule. Going along a row from left to right, the reactivity increases, and properties such as electronegativity increase as well. Going downward, the reactivity decreases. Other properties vary differently from the column-wise view of the periodic table of elements. The greater the ratio, the greater the probability that the element will be radioactive. If the ratio is greater than 1, the probability increases that an element (or an isotope of an element) will be radioactive and unstable. The ratio of protons to neutrons is important in determining the stability of elements. They contain elements such as plutonium, radon and uranium, and, notorious since the poisoning scandal in the UK, polonium. These instabilities lead to radioactive decay as is evident from the actinides and lanthanides, the groups of the rare earths. The stability of these elements diminishes however with an increasing number of nucleons (protons and neutrons). Silicon (Si) responds in the same way and silicon chemistry is to a significant extent similar to carbon chemistry.Įlements in a row show different periodicities – such as electronegativities, increasing atomic mass, increasing number of protons and neutrons. The bonds of the carbon (C) take a tetrahedral or pyramidal shape. For instance carbon and hydrogen (H 2) react to form methane. The way the element at the top of a column reacts and the way it produces new chemicals give a clue to how other elements in that same group (column) will react. As a result, the likelihood of reactants to be able to react together can be determined from the Periodic Table of Elements. In all these metals there is only one free electron and these ball-shaped orbitals are denoted by 1 s, 2 s, 3 s, etc., all with an increasing average radius from the nucleus.įor all the subsequent groups, characterizations can be formulated based upon similarities in reactivity-the way they react, how many electrons can be shared, etc. The one free electron in the outer valence shell is in a so-called ns- orbital. Because these electrons are all at a greater distance from the nucleus, the energy gained from removing one electron diminishes according to the proportionality of n −2. As the mass of the nucleus increases with each neutron and proton, so also does the number of electrons (to balance the electric charge). This lowered reactivity is related to the atomic weight. Going down from Li, Na, K to Fr the reactivity decreases, but these metals react easily – for example, with water, in a very exothermic way. They are characterized by having a single electron in their outer valence orbital. On the extreme left there is a group of metals, called the alkali metals. (See the article atomic orbital for a more detailed explanation of the building up of the electronic shells.) 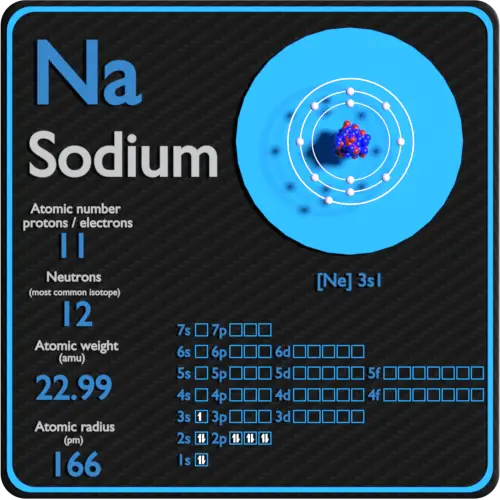
Because they have an outer shell of electrons that is completely filled, they are inert in behavior. For instance, the right column is occupied by the noble gases. The number of electrons in the outer shells of the electron orbitals is the same within a group, only the principal quantum number n describing the orbitals increases. Elements by periodic table group (vertical column) See Atomic electron configuration for the orbital occupancies of ground state atoms.Įlements in any one group behave in a similar way and show the same overall general properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
